 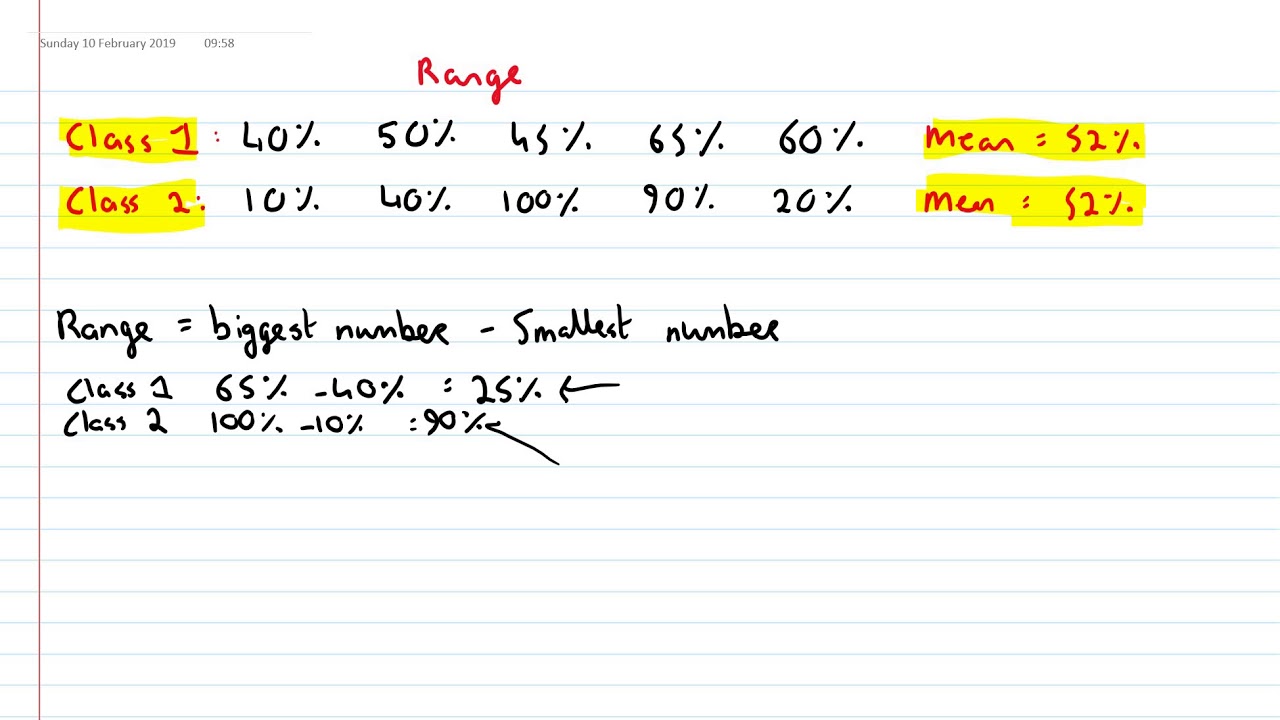
Cells from all depots are represented in the U and L branches (the percentage of cells from each depot is indicated for each branch). Following induction of differentiation, the P branch split into an upper (U) and a lower (L) branch, thus containing cells from T4 and T5. Cells from T1–T3 align in a common progenitor (P) branch. d, Monocle pseudotime trajectory of adipocyte progenitors from T1–T5. Clustering analysis grouped T1–T3 samples by adipose depot and T4 and T5 samples by time point and adipose depot. 
The inset indicates the collection time point. c, t-SNE atlas of developing adipocyte progenitors (T1–T5) of four adipose depots from different individuals. Peri, perirenal subq, subcutaneous supra, supraclavicular visce, visceral. b, t-SNE atlas generated using the Seurat alignment algorithm, analysing proliferating adipocyte progenitors (T1) derived from 11 individuals. a, Overview of adipose depots and cellular developmental stages. Human adipocyte progenitors isolated from tissue biopsies of four adipose depots were collected at five time points (T1–T5) during in vitro differentiation and subsequent single-cell analysis was performed using the 10x Genomics platform. 
Gene expression plots for our data can be visualized using the web tool available at. Finally, we confirm that all these cell types are present in vivo using machine learning-based label transfer algorithms. We study these cells during early differentiation and combine computational modelling with experimental cell separation techniques to describe two cell-type branches that arise from a common progenitor. In the current study, we compared human adipose stem and progenitor cells (hASPCs) derived from two WAT depots-subcutaneous and visceral-and from two BAT depots-supraclavicular and perirenal. However, the developmental hierarchical heterogeneity of human brown and white adipogenic events remains elusive. These studies suggest that separate cell types provide thermogenesis, insulin sensitivity, lipid storage and adipokine secretion 9, 10, 11 or act as negative regulators of lipid accumulation 12. Single-cell technologies have allowed an understanding of the heterogeneity of adipocytes, revealing several subtypes with specialized functions 6, 7, 8. Intriguingly, progenitor cells derived from the seasonally plastic white adipose tissue (WAT) of brown bears spontaneously differentiated into osteocytes in vitro 5, further emphasizing the developmental link between these two cell types. Other studies have shown a close relation between osteocytes and adipocytes, with opposing differentiation trajectories mediated through a common transcriptional network 4. Adipocytes originate from mesenchymal stem cells that reside in multiple tissues including adipose tissue, skeletal muscle and bone marrow 3. Brown adipose tissue (BAT) activity is also associated with metabolic health 2. Multiple studies have described that abdominal obesity is strongly associated with cardiovascular disease and insulin resistance, whereas accumulation of fat in the lower gynoid regions has a lower lipid turnover and is associated with metabolic health 1. In conclusion, we provide a differentiation map of human adipocytes and define the multipotent SWAT cell, providing a new perspective on adipose tissue regulation.īody fat distribution and adipocyte functionality are determinants of metabolic health in a depot-dependent fashion. Label transfer algorithms recapitulate the cell types in human adipose tissue datasets. When stripped from adipogenic cells, SWAT cells display a multipotent phenotype by reverting towards progenitor state or differentiating into new adipogenic cells, dependent on media. 
Based on an extracellular matrix and developmental gene signature, we name the structural branch of cells structural Wnt-regulated adipose tissue-resident (SWAT) cells. The adipogenic gene signature contains mitochondrial activity genes, and associates with genome-wide association study traits for fat distribution. We show at single-cell resolution that progenitor cells from four human brown and white adipose depots separate into two main cell fates, an adipogenic and a structural branch, developing from a common progenitor. Adipocyte function is a major determinant of metabolic disease, warranting investigations of regulating mechanisms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
